|
1/18/2024 0 Comments Atomic spectra meaning 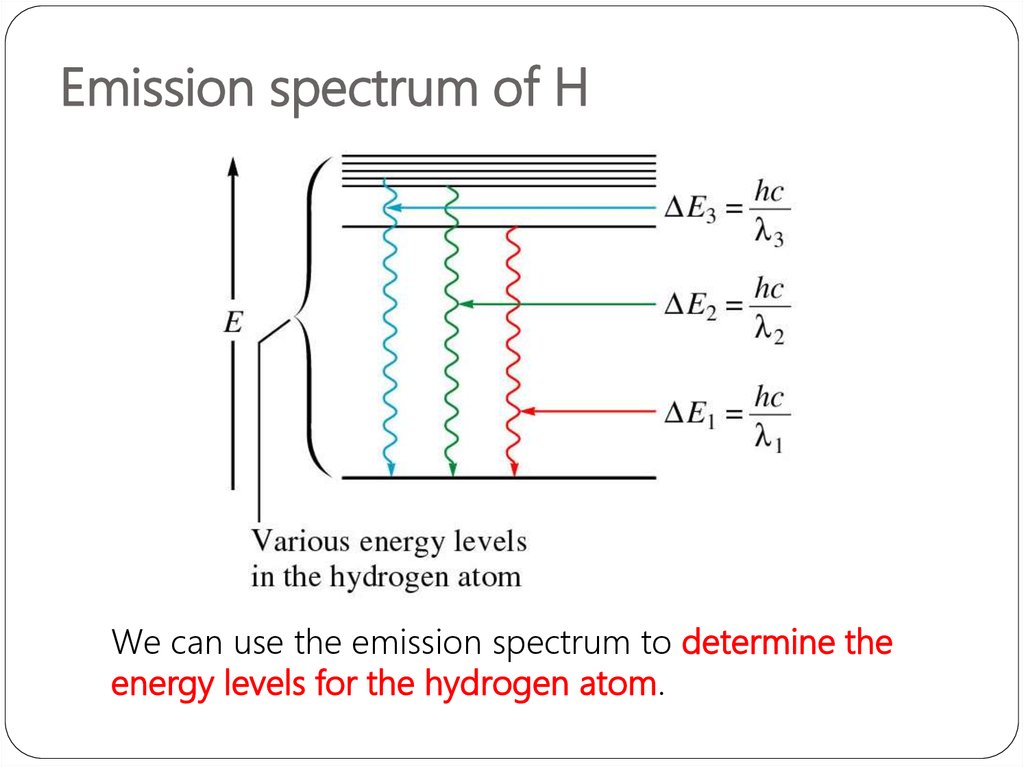
The wavelengths, intensities, and spectrum assignments are given in. The compilation includes data for the neutral and singly-ionized atoms of all elements hydrogen through einsteinium ( Z 1-99). The expression forbidden transitions is often used, but this does not mean that these transitions cannot occur, only that they are electric-dipole-forbidden. Define the ground state ground state and excited state of electrons Explain the difference between line spectra for absorption and emission of electrons. This handbook is designed to provide a selection of the most important and frequently used atomic spectroscopic data in an easily accessible format. These considerations generate different sets of transitions rules depending on the multipole order and type. Thus, parity does not change for E-even or M-odd multipoles, while it changes for E-odd or M-even multipoles. In quantum mechanics the basis for a spectroscopic selection rule is the value of the transition moment integral ∫ ψ 1 ∗ μ ψ 2 d τ, Evidence from atomic line spectra supports the Bohr model of the atom using photon frequency calculations. Spectral lines are often used to identify atoms and molecules. Spectra All elements either emit or absorb certain frequencies of light. In the following, mainly atomic and molecular transitions are considered. A spectral line is a dark or bright line in an otherwise uniform and continuous spectrum, resulting from the emission or absorption of light in a narrow frequency range, compared with the nearby frequencies. The selection rule also plays a role in chemical reactions, where some are formally spin-forbidden reactions, that is, reactions where the spin state changes at least once from reactants to products. The selection rules may differ according to the technique used to observe the transition. 
Selection rules have been derived for electromagnetic transitions in molecules, in atoms, in atomic nuclei, and so on. Because they are unique, they can act as an element s fingerprint. In physics and chemistry, a selection rule, or transition rule, formally constrains the possible transitions of a system from one quantum state to another. noun : a spectrum of radiation due to electron transitions within atoms and consisting mainly of series of spectrum lines characteristic of the element Love words You must there are over 200,000 words in our free online dictionary, but you are looking for one that’s only in the Merriam-Webster Unabridged Dictionary. Atomic emission spectra are unique spectra of light emitted by an element when electricity is run through it or when it is viewed through a prism. Formal constraint on the possible transitions of a system from one quantum state to another 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
